Message from the President
 Representative Director, President
Representative Director, President
Takao Ota
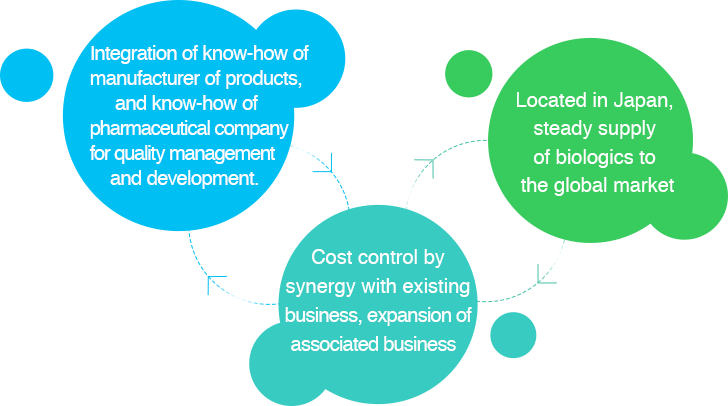
The market of biologics including antibody drugs has been rapidly expanding in recent years as they are expected to perform superior effectiveness and safeness compared to conventional low-molecular drugs. Meanwhile, considerable capital investment and running cost are required to manufacture antibody drugs as both high level expertise and dedicated equipment are indispensable. It has become a challenge to secure an appropriate facility and manufacturing scale to meet the demand.
We established Cultivecs Inc. to tackle these issues. Cultivecs constructed a GMP-compliant antibodies manufacturing facility in Niigata, Japan, and launched operations in 2018. At this facility, we put every our effort to produce top-quality antibdies at lower cost, in addition to that trying to maintain close communication, fine-tuned support and accurate delivery.
We would like to ask for your continued support and encouragement.
Mission
We make contribution to control medical expenditures and development of medical technology through manufacturing of high quality and low cost antibody drugs by state-of-the-art technology.